Streptococcus pneumoniae
The IHME estimates that Streptococcus pneumoniae (S. pneumoniae) is responsible for ~1.2 million deaths and ~900,000 years lived with disability annually from pneumonia and meningitis. In addition, S. pneumoniae is responsible for ~35% of the global burden of pneumonia and ~25% of the global burden of otitis media. Effective vaccines, including PCV13 (Prevnar), are available and have excellent efficacy but global coverage remains low at ~40%, primarily due to high cost per dose. Coverage is particularly low in India and China. By the end of 2016, Gavi had approved support for PCV introduction in 59 countries and plans to extend support. Current R&D aims to develop lower cost platforms and manufacturing, as well as to increase serotype coverage.
S. pneumoniae falls into a cluster of pathogens for which the priority is to increase vaccine uptake. The primary recommendation is to drive coverage and equity. The secondary recommendation is to support pre-clinical research.
S. pneumoniae is a Gram-positive bacterium often found as a commensal in the upper respiratory tract. Globally, most children will acquire S. pneumoniae in their nasopharynx in early life 412–416. S. pneumoniae is transmitted through contact with respiratory droplets from patients or carriers. The most common manifestations are pneumonia and otitis media, but S. pneumoniae infection can also result in meningitis and sinusitis. Symptoms of pneumonia caused by S. pneumoniae include fever, chest pain, cough, rusty sputum, dyspnoea, tachypnoea/tachycardia, and hypoxia; otitis media caused by S. pneumonia manifests as ear pain, hearing difficulties, swollen ear drum, and fever.
Groups at risk of S. pneumonia infection include children under 5 years, particularly those under two years, and adults over 65 years. S. pneumoniae has a global distribution; however, in 2015 half of global pneumococcal deaths occurred in only four countries in Africa and Asia (India, Nigeria, the Democratic Republic of the Congo, and Pakistan) 417.
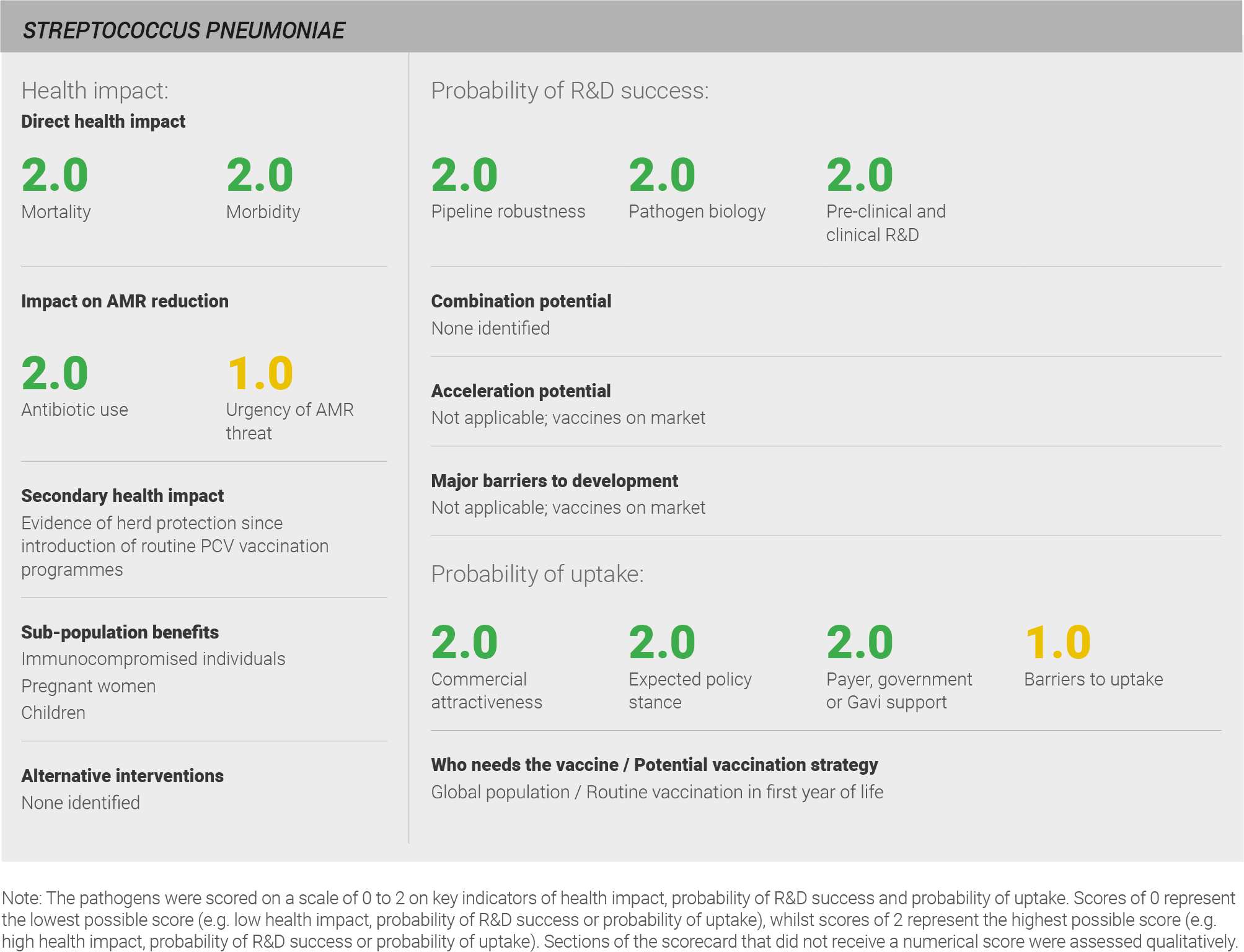
Direct health impact
Robust global data on disease burden related to all S. pneumoniae-associated infections is not available. IHME estimates for mortality and morbidity from pneumococcal meningitis and pneumococcal pneumonia are available for 2016. In 2016, pneumococcal meningitis was estimated to be responsible for 23,000 deaths and 600,000 years lived with disability and pneumococcal pneumonia was estimated to be responsible for 300,000 deaths and 1.2 million years lived with disability 31. No robust global data is available for otitis media but a review of the literature suggests that S. pneumoniae is responsible for 27% of acute otitis media globally 418. Taken together, this evidence suggests that S. pneumoniae infection causes significant health impact.
As mentioned, no publications report total mortality and morbidity for this pathogen, but S. pneumoniae mortality and morbidity from meningitis and pneumonia are reported by the IHME. This data source has an accepted methodology and is used in the global health community. In this assessment, IHME morbidity estimates were combined with estimates for the percentage of otitis media caused by S. pneumoniae infection taken from the literature. This estimate may be less precise than the IHME estimate. A full methodology for this assessment can be found in the appendix.
Scoring: Based on the above analysis, mortality was categorised as high (score of 2 out of 2) and morbidity was categorised as high (score of 2 out of 2).
Secondary health impact
There is significant evidence of herd protection for S. pneumoniae. Vulnerable populations, including the elderly, benefit from the vaccination of infants, who are the primary reservoir of S. pneumoniae 419.
Sub-population benefits
Vaccines against S. pneumoniae particularly benefit children, pregnant women, and immunocompromised populations, including those with HIV.
Antibiotic use
Typical first-line antibiotic treatment includes beta-lactams and cephalosporins 420. The treatment course varies depending on the specific condition; a typical course of antibiotics is seven days, but treatment for meningitis is usually longer. Antibiotic use is driven by acute otitis media and lower respiratory tract infection as these are the most common S. pneumoniae infections. The widespread use of antibiotics to treat these infections makes S. pneumoniae an attractive target for broader vaccine coverage, as one expert notes “the vaccine that would have the highest impact on antibiotic use would be S. pneumoniae”28.
Scoring: Based on the above analysis, antibiotic use was categorised as high (score of 2 out of 2). This estimate is based on an annual incidence of ~120 million LRTIs treated with a one week course of antibiotics, ~0.5 million meningitis cases treated with a two-week course of antibiotics and ~ 120 million acute otitis media cases treated with a one week course of antibiotics.
Urgency of AMR threat
The WHO and CDC have both expressed concern about the future of S. pneumoniae treatment. S. pneumoniae is listed as “medium” in the WHO priority list of R&D for new antibiotics and listed as a “serious” threat in the CDC list of biggest threats from AMR. Resistance to first-line penicillin varies by region from approximately 2% in the United States to up to 70% in Vietnam 421,422. In 30% of severe cases, S. pneumoniae is fully resistant to one or more clinically relevant antibiotics 7.
Scoring: Based on the above analysis, the urgency of AMR threat was categorised as medium (score 1 out of 2).
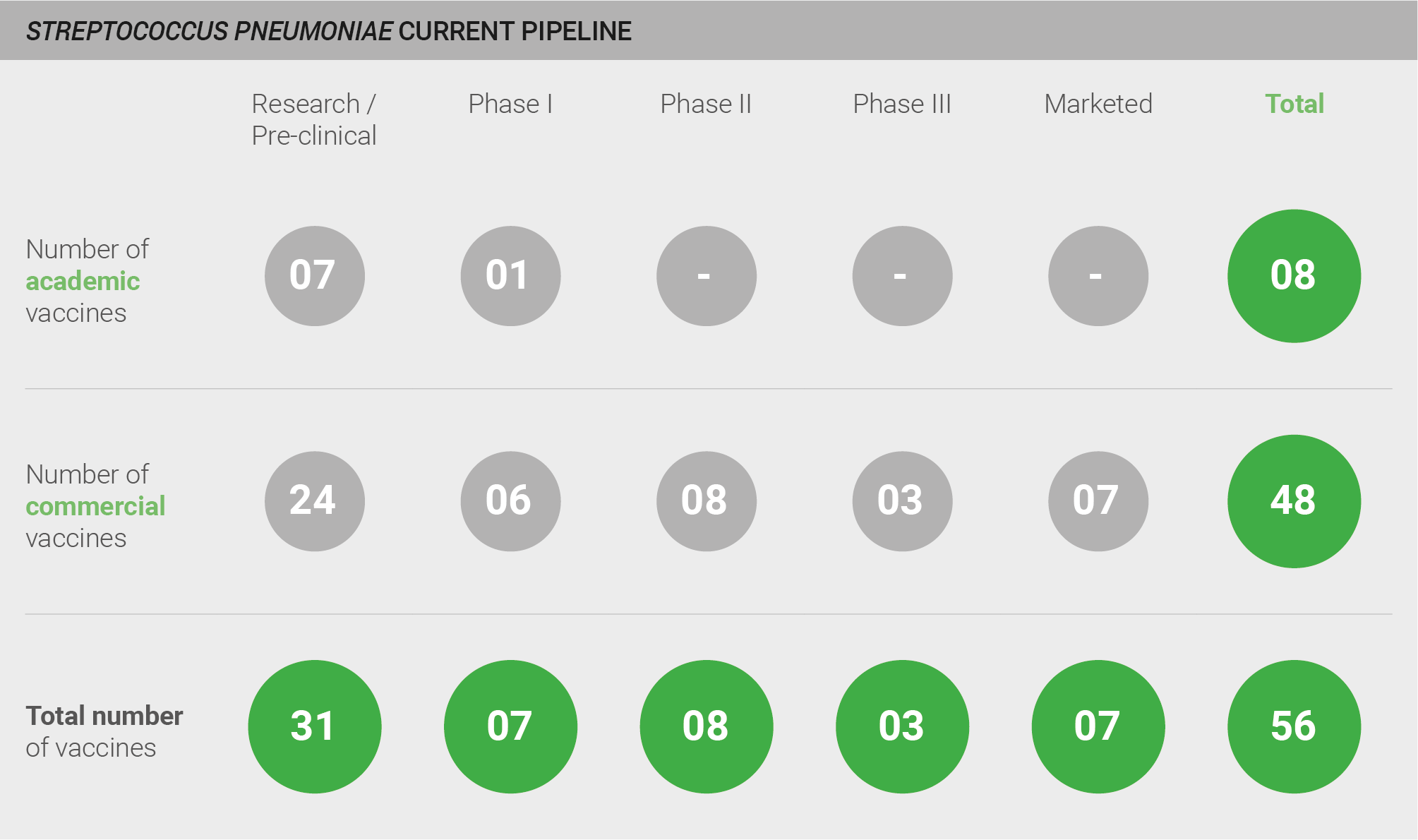
Pipeline robustness
The pipeline for development of vaccines against S. pneumoniae is robust, comprising a total of 56 vaccines, including seven marketed vaccines. Those still in development include 31 vaccines in pre-clinical studies, seven in Phase I, eight in Phase II, and three in Phase III.
Scoring: Based on the above analysis, pipeline robustness was categorised as high (score of 2 out of 2).
Current pipeline
Profile and impact of current vaccines
The marketed vaccine PCV13 (Prevnar) provides a precedent for S. pneumoniae vaccine development. The overall vaccine efficacy of ≥1 dose of the 13 valent pneumococcal conjugate vaccine (PCV13) for preventing invasive pneumococcal disease due to vaccine serotypes is ~76% 423. Overall vaccine efficacy is estimated to be 90% when ≥2 doses are given before 12 months of age, two doses are given on or after 12 months of age, or one dose is given on or after 24 months of age 423.
Whilst PCV13 is highly effective, in areas where the vaccine is widely used non-vaccine serotypes of S. pneumoniae increase in prevalence to fill the ecological niche that has been vacated by vaccine serotypes 424. Experts acknowledge this, stating “we are seeing some serotype shifting in response to Prevnar and the challenge is to continue to expand coverage against additional serotypes” 28. However, as noted by another expert, “[the] more virulent strains are the ones we try to include in our vaccine,” 28. Thus, the strains that increase in prevalence in response to vaccination may be less virulent.
Pathogen biology
Partial strain-specific natural immunity to S. pneumoniae is known to be possible 425–427. However, the immune response to S. pneumoniae infection is still not entirely understood. It appears to be complex and multi-layered, and defence mechanisms include both cellular and secreted components of the immune system 425–427.
Vaccine targets for S. pneumoniae are well-characterised. Polysaccharides are established targets with a decades-long history of effective use in S. pneumoniae vaccines 428. Conjugated vaccines (polysaccharides covalently bound to diphtheria toxoid) induce a more robust and long-lasting immune response 429. Existing vaccines target a range of specific S. pneumoniae serotypes. However, a conserved antigen-based vaccine capable of inducing cross-strain immunity would address serotype shifts but has not yet been developed 428,430.
Scoring: Based on the above analysis, pathogen biology was categorised as high (score of 2 out of 2).
Pre-clinical and clinical R&D
Animal models currently in use for pre-clinical S. pneumoniae research are well-established and have solid predictive capacity for clinical programmes. Characteristics of pneumococcal pneumonia, sepsis, and meningitis have successfully been reproduced in mice, rats, and rabbits 431. Models for otitis media are also available and include the chinchilla 432, gerbil 433, and rat 434. All of these models continue to be helpful tools in elucidating aspects of disease pathogenesis, characterising innate and adaptive immune responses to S. pneumoniae, and testing the efficacy of antibiotics and other therapies, as well as potential vaccine candidates. Pre-clinical research is also facilitated by the identification of correlates of protection for S. pneumoniae 435.
Clinical research and development benefits substantially from prior vaccine development for S. pneumoniae. Intranasal challenge studies are possible for S. pneumoniae 436, and trial infrastructure is in place. Efficacy trials for vaccines have been conducted previously, as have effectiveness studies 417. However, some limitations do exist; whilst correlates of protection have been identified, serotype-specific correlates of protection vary widely 437. Also, the relationship between IgG concentration after priming and long-term protection needs to be better understood 438. Finally, diagnostics for surveillance still rely heavily on insensitive culture techniques, and new methods such as PCR and proteomics are needed.
Scoring: Based on the above analysis, pre-clinical and clinical R&D was categorised as high (score of 2 out of 2).
Expected policy stance
The target population for vaccination is infants and the vaccination is included in the routine vaccination schedule. The WHO recommends three primary doses, or two primary doses and a booster. The WHO recommends the inclusion of PCV as a priority in childhood immunisation programmes worldwide, particularly in countries with high mortality in children under 5 (>50/1000 live births) 439.
Scoring: Based on the above analysis, expected policy stance was categorised as high (score of 2 out of 2).
Payer, government, or Gavi support
Conjugated PCV vaccines are included in routine vaccination schedules in high- and middle-income countries and in some low-income countries. More than 50 Gavi countries have introduced PCV vaccines into their routine programmes 440. In 2007 Gavi announced a pilot Advance Market Commitment (AMC) for funding these vaccines. The AMC was officially launched in 2009 and has helped result in coverage for Gavi countries being similar to the global average 404,440,441. However, there is a lack of support in some middle-income countries, particularly in India and China. Both countries have PCV vaccination rates below 10% 408,409. In China, PCV has not been integrated into the Chinese Expanded Program on Immunization, so individuals have to pay for pneumococcal vaccination themselves 442. In India, local manufacturing at scale is likely to emerge. The Serum Institute of India is developing a 10-valent vaccine 411. In both countries, in-country development of production capabilities for PCVs is ongoing, which will likely provide vaccines at a lower price point.
Scoring: Based on the analysis described above, payer, government, or Gavi support was categorised as high (score of 2 out of 2).
Barriers to uptake
The high price point of marketed S. pneumoniae vaccines has been a key challenge to expanding vaccination coverage. Current coverage is estimated at ~40% worldwide 404. This is heavily driven by the high cost of the vaccine, which affects low-income countries and those “graduating” from Gavi support. According to one expert “lower cost multivalent vaccines could make a huge difference in uptake” 28. For example, the PCV13 vaccine is produced using an established method, production costs are comparatively high and globally produced quantities are still low. There is a trend toward more cost-efficient production and the Serum Institute of India is collaborating with PATH to develop an affordable 10-valent PCV, focusing on the serotypes prevalent in 70% of the affected population (Asia, Africa, LAC, India) 411.
S. pneumoniae vaccination uses established vaccination touchpoints 1. However, some logistical challenges to implementing a vaccination programme for S. pneumoniae exist. In addition to the cost of the vaccine, lack of evidence regarding the country-specific burden of S. pneumoniae infection and lack of local expertise in economic evaluation contribute to low current coverage rates. Child outmigration, travel distance to healthcare centres, low maternal education and low socio-economic status are also associated with reduced uptake 443. Also, many countries do not have effective catch-up campaigns in place 407.
Scoring: Based on the above analysis, barriers to uptake was characterised as medium (score of 1 out of 2).
Commercial attractiveness
In this assessment, commercial attractiveness has been categorised as high, reflecting the commercial success of S. pneumoniae vaccines. Overall, the global pneumococcal vaccine market size was estimated to be $7 billion in 2017 444.
Scoring: Based on the above analysis, commercial attractiveness was characterised as high (score of 2 out of 2).
Primary recommendation
The primary recommendation is to drive equity and coverage for S. pneumoniae vaccination. Current coverage in India and China is particularly low. In India, efforts should be made to accelerate local production and to invest in infrastructure for delivery. Ensuring better delivery is likely to increase coverage, as 62% of children aged 12-23 months received all basic vaccinations in 2015-2016 – a substantially higher proportion than those currently vaccinated against S. pneumoniae. In China, local production should be developed, and the inclusion of vaccination against S. pneumoniae in the Expanded Program on Immunisation schedule should be explored. This would allow in-country manufacturers to plan vaccine production on a more secure demand level.
Secondary recommendation
The secondary recommendation is to invest in pre-clinical research. Whilst marketed vaccines exist, pre-clinical research could improve on existing vaccines and facilitate the development of vaccines that are cheaper to produce. Additional pre-clinical research could also address serotype shifts resulting from vaccine pressure by facilitating development of a conserved antigen-based vaccine that can induce cross-strain immunity; such a vaccine has not been developed to date. Pre-clinical research should also continue to support serotype expansion and serotype replacement, given the variation in serotype distribution across geographies. Finally, serotype-specific correlates of protection vary widely, and the relationship between IgG concentration after priming and long-term protection needs to be better understood.